📌 Course Overview
The Malaysian Standard MS 1480:2025 is the third revision of the HACCP standard and officially replaces MS 1480:2019. This revision introduces clearer structure, enhanced hazard analysis expectations, expanded validation and verification requirements, strengthened documentation controls, and new clauses related to operation, maintenance and continual improvement of the HACCP system.
This HACCP MS1480 Transition Training (2019 → 2025) is specifically designed to guide organisations through the transition process by explaining what has changed, why it has changed, and how to comply with the latest standard.
📌 Official transition reminder (ACB Circular 1/2026 – Issue date: 20 Jan 2026):
• JSM to witness CB based on MS 1480:2025 no later than: 1 Oct 2026
• CBs to complete transition of all clients by: 24 Oct 2027
• After 24 Oct 2027, certification to MS 1480:2019 will no longer be accepted
• (Also noted: HACCP elements in FSMS should be based on MS 1480 to be eligible for Health Certificate for Export)
The training provides both technical understanding and practical application, enabling participants to update existing HACCP systems in line with MS1480:2025 requirements.
🎯 Learning Objectives
By the end of this training, participants will be able to:
- Understand the key differences between MS1480:2019 and MS1480:2025
- Identify new and revised requirements impacting HACCP systems
- Apply updated hazard analysis and CCP determination methods
- Strengthen validation, monitoring and verification practices
- Update HACCP documentation and records to meet audit expectations
- Prepare organisations for MS1480:2025 transition audits
👥 Target Participants
- HACCP Team Members
- HACCP Team Leaders / Coordinators
- Quality Assurance & Quality Control Personnel
- Production Managers & Supervisors
- Regulatory & Compliance Officers
- Food Safety Executives
📚 Key Topics Covered
- Overview of MS1480 revision (2019 → 2025)
- Clause-by-clause comparison of major changes
- Updated product description and process flow diagram requirements
- Enhanced hazard analysis methodology and acceptable hazard levels
- Validation and verification scope expansion
- Monitoring system improvements and trend analysis
- New requirements for emergency preparedness & response
- Control of measuring equipment and methods
- HACCP documentation, record-keeping and system improvement
⏱ Duration & Method
-
Duration: 1 Day (7 Hours)
-
Method: Lecture, discussion, practical examples
-
Language: English & Bahasa
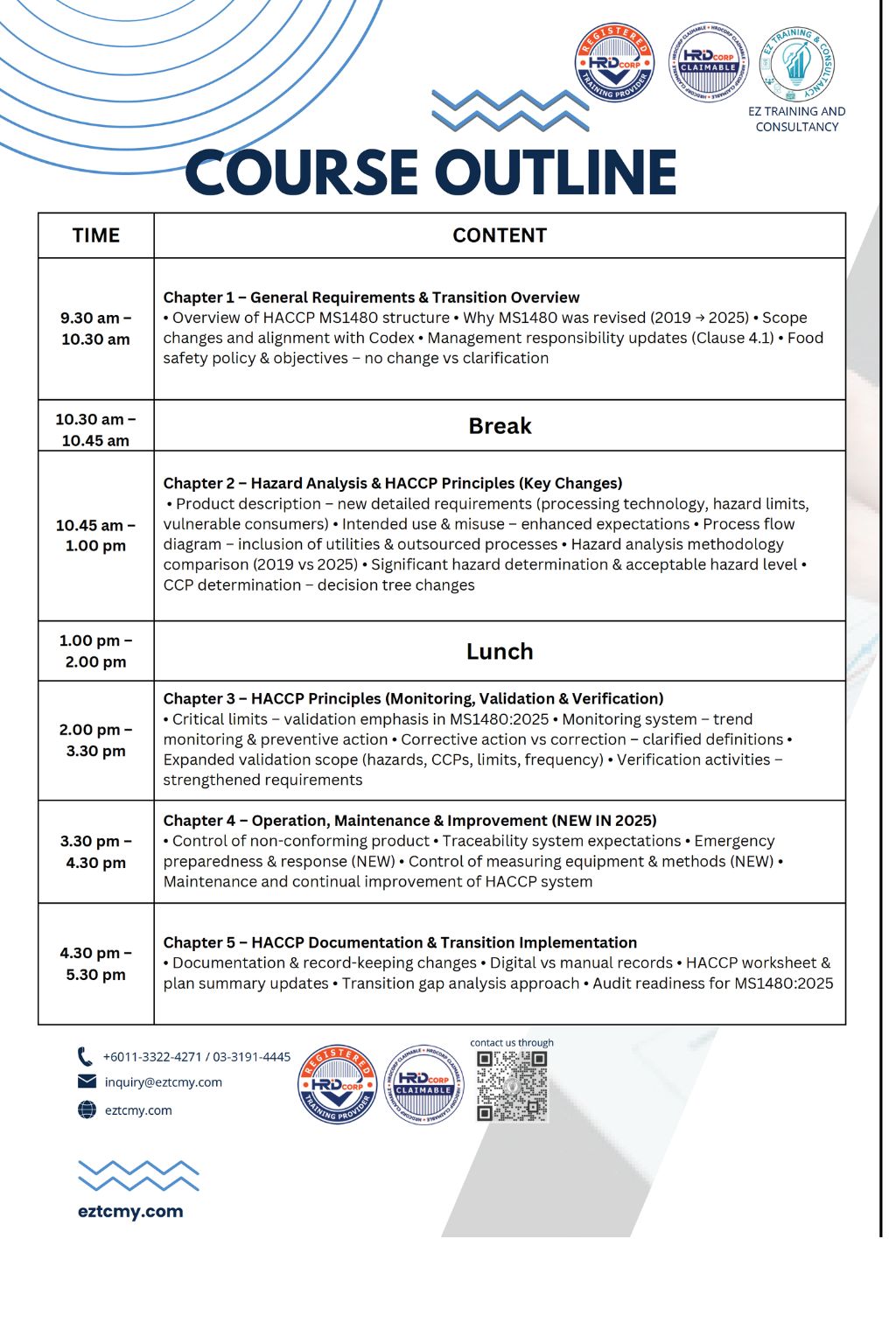
TIME CONTENT 9.30 am – 10.30 am Chapter 1 – General Requirements & Transition Overview • Overview of HACCP MS1480 structure • Why MS1480 was revised (2019 → 2025) • Scope changes and alignment with Codex • Management responsibility updates (Clause 4.1) • Food safety policy & objectives – no change vs clarification 10.30 am – 10.45 am Break 10.45 am – 1.00 pm Chapter 2 – Hazard Analysis & HACCP Principles (Key Changes) • Product description – new detailed requirements (processing technology, hazard limits, vulnerable consumers) • Intended use & misuse – enhanced expectations • Process flow diagram – inclusion of utilities & outsourced processes • Hazard analysis methodology comparison (2019 vs 2025) • Significant hazard determination & acceptable hazard level • CCP determination – decision tree changes 1.00 pm – 2.00 pm Lunch 2.00 pm – 3.30 pm Chapter 3 – HACCP Principles (Monitoring, Validation & Verification) • Critical limits – validation emphasis in MS1480:2025 • Monitoring system – trend monitoring & preventive action • Corrective action vs correction – clarified definitions • Expanded validation scope (hazards, CCPs, limits, frequency) • Verification activities – strengthened requirements 3.30 pm – 4.30 pm Chapter 4 – Operation, Maintenance & Improvement (NEW IN 2025) • Control of non-conforming product • Traceability system expectations • Emergency preparedness & response (NEW) • Control of measuring equipment & methods (NEW) • Maintenance and continual improvement of HACCP system 4.30 pm – 5.30 pm Chapter 5 – HACCP Documentation & Transition Implementation • Documentation & record-keeping changes • Digital vs manual records • HACCP worksheet & plan summary updates • Transition gap analysis approach • Audit readiness for MS1480:2025







Reviews
There are no reviews yet.